 Over 100 registrations and 6 speakers from industry and academia exchanged experiences during the online 10th IGC Symposium, 15th June 2021. Topics were surface properties of catalysts (DSM), coated plates (Wicoatec), cement (EPFL), novel packaging (Univ. of Alberta), membranes (Adscientis) and more – measured by dynamic gas adsorption, also known as Inverse Gas Chromatography (IGC). The very positive feedback of the participants and the numerous connections afterwards on different topics confirmed the format: science-based IGC with industrial relevance and the openness to discuss also controversial topics! Thought-provoking was definitely Steven Abbott’s presentation about his new concept of “Surface Interaction Parameters”, in short SIP. It is based on the idea of a normalized adsorption energy and inspired by the HSP-concept. How are adsorption isotherms and measurements at very low, infinite dilutions are connected? Nicolas Leborgne, Adscientis, explained not only, how adsorption isotherms of two different solvents are efficiently obtained by IGC-FC (finite concentration), but also how the different results complement each other. The Adsorption Energy Distribution Function (AEDF) describes the higher (and lower) energy sites – and these results are congruent to the results at infinite dilution. Furthermore, this talk sparked a deeper exchange about the interpretation of adsorption isotherms – great! The high sensitivity of the 15+ molecular probes used in IGC was demonstrated by Jan Schütz, DSM, by the example of catalyst deactivation at different bed locations and the detailed description about the changes in surface properties. The high sensitivity of IGC was also key in Franco Zunino’s presentation. His research at EPFL on “greener” cement requires detailed insights about materials, treatments and reactivities. IGC delivered exactly the surface sensitive information in addition to images and XRD characterizations. The growing IGC community was further demonstrated by the international partners of Adscientis, namely Dhananjay Kulkarni (LabIndia, India), Zhi Ouyang (Gravite, China) and Joe Domingue (SciMark International, US). Surely, this online IGC Symposium brought many different minds together and triggered many thoughts - a great event to remember! Note: most of the presentations are available at the Inolytix Youtube channel, see link.
0 Comments
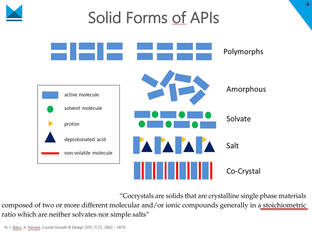 The background: one of the major challenges in pharmaceutical formulation is the low solubility of APIs. A very elegant, but non-trivial way to improve or even control the solubility and therefore bioavailability of an active compound is the formation of a cocrystal. Combined with non-volatile coformer molecules, the API forms a new crystalline single phase and thereby creates a new material that is different from simple polymorphs or physical mixtures of the API and conventional formulation ingredients. Thus cocrystals may also offer interesting opportunities to circumvent patent issues. Nevertheless, up to now finding a suitable cocrystal usually requires expert knowledge and a vast variety of experiments. The speaker Carsten Schauerte, with over 20 years of experience in the field, presented a new method to improve the conventional screening approach by a goal-oriented way of using ternary phase diagrams prior to and in combination with the actual screening procedure. The example of carbamazepine with different conformers and solvents explained the approach. Cocrystallisation offers many advantages, but the complexity and difficulties of cocrystal- screening should not be underestimated. An experienced partner as SOLID-CHEM could significantly save time and open the window for new opportunities. Please contact us for any enquiries:
Please note: the link to the recorded webinar is available on request. Graphenes, single and multiwall carbon nanotubes (CNTs) and many different applications from batteries to conducting composites were topic of this NanoCarbon annual conference initiated by the German Network NanoCarbon and the Nanoinitiative Bayern GmbH. Started in 2015, this conference has become more and more international and with 34 presentations, interactive “coffee rooms” and lively discussions after presentations, the new online format has been proven successful over the two day, 02.-03.03.2021. My presentation “surface properties of graphenes, carbon blacks and carbon fibers by IGC” received a great deal of interest. No wonder, interaction behavior and surface functionalization was often mentioned, but analysis has been mostly restricted to SEM or TEM images or application results. We are now in contact with several institutes and are looking forward to contribute towards the many different applications. The abstract is attached below. The complete set of slides can be requested by the author, just send an email to [email protected].
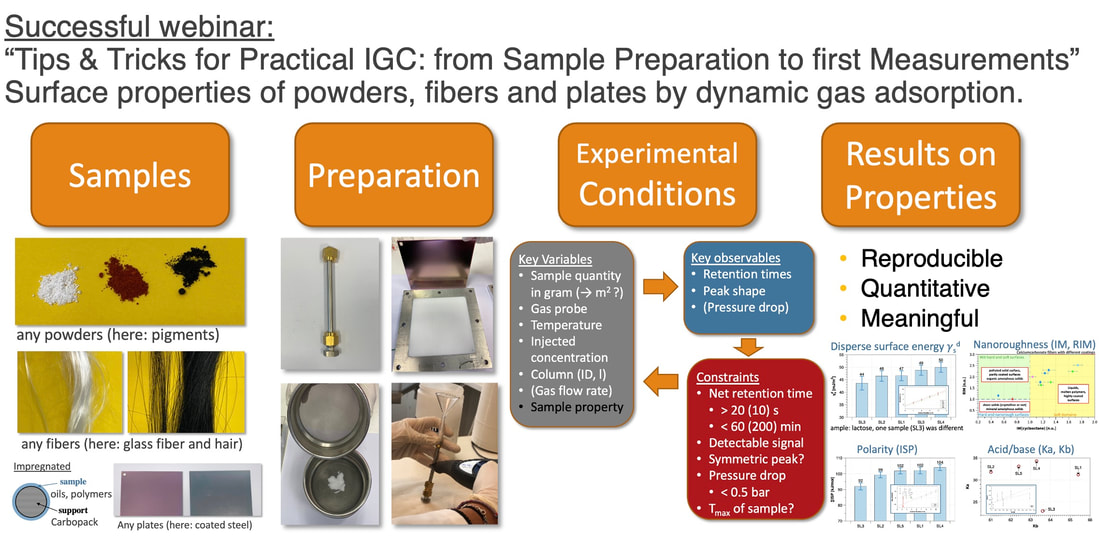
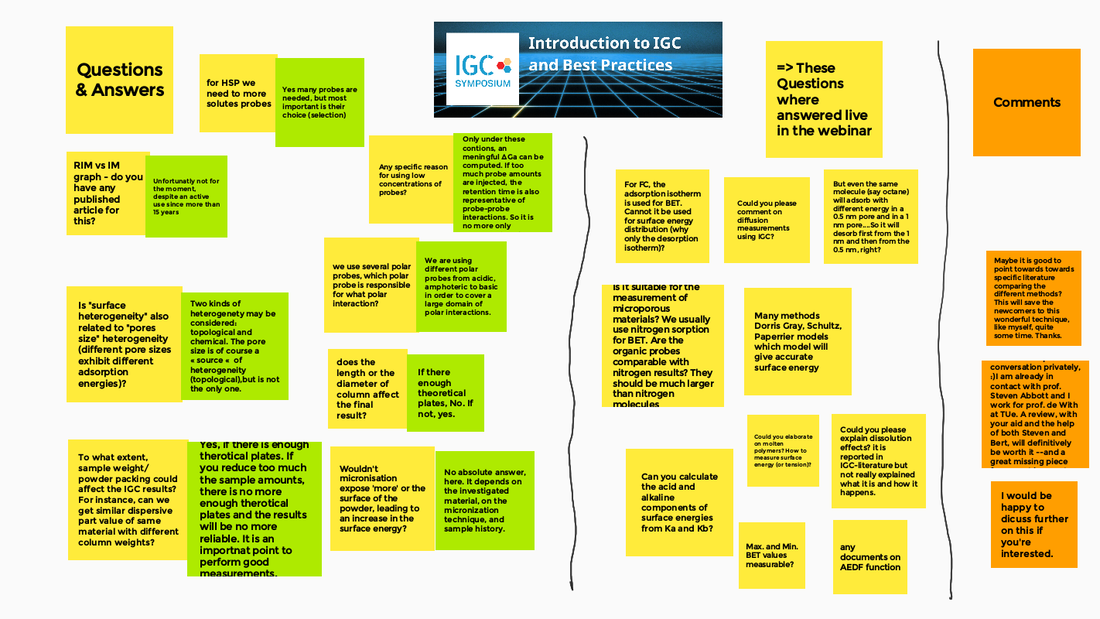
2nd Webinar in our series "IGC explained"On the 10th February we held another webinar about IGC with more than 60 registrations from Europe but also from India, China, Egypt and Canada which was a great surprise for us. In this webinar we presented and explained step-by-step the sample preparation, choosing, how to fill column properly and how to run first experiments with alkanes and polar probes. We also gave practical tips how to find the appropriate condition of sample weight, flow rate and temperature. Other points included to minimize the pressure drop with nano-scale material, how to use fibrous material or even how to use a shorter column for polar probes and a longer one for non-polar probes – and combine the data. You find a graphical summary below or you may even watch the recording of the webinar on our Youtube channel, see linke below. If you have any questions concerning IGC or any other method for surface characterization, feel free to contact us.  This article is about our partner SOLID-CHEM GmbH, its beginnings and why it has received the Innovation Medal, or in German “Stiftersiegel,” for Innovation by Research awarded by the German Donor’s Association (Stifterverband)[1] for its intensive and dedicated activities in research. The Beginning in 2010 SOLID-CHEM has its origin as a spin-off from University Duisburg-Essen, founded in 2010 at the Bochum University Campus by the two enthusiastic scientists Professor Dr. R. Boese and Dr. C. Schauerte. “About three decades ago,” Boese recalls, “crystal engineering came up as part of solid-state chemistry and gained importance in organic and pharmaceutical Industry.” This was obvious with an increasing number of patent applications and litigations on polymorphism of APIs (Active Pharmaceutical Ingredients) and the request for experts in the field. Our research interests originated in crystallization techniques in crystallography but gradually shifted to co-crystals, salts, amorphous phases, and the characterization in the wide field of analytic techniques, - spectroscopic, diffraction, thermochemical, particle size determinations, etc. Competences and early Challenges About ten years ago, they had an extensive network to Industry. With the desire for more independence and space, we recognized that all this would fit for a new business, so we looked for suited laboratories and basic lab equipment. Focused on material properties like solubility, bioavailability, melting temperature, shelf-life stability, hygroscopicity, filtering properties, compressibility, tableting ability, and so on, we were in the perfect position for recruiting our team from the previous research group at University. It appeared that we had to face many new challenges, such as organizing the internal structure and always having economic needs in vision. Carsten Schauerte was fortunate to spot suited laboratories right in time at a new-built technology center next to the Bochum Ruhr-University campus. “This was an ideal environment for us, with contacts, support for equipment and scientific environment, with the vicinity for discussions and collaboration, with numerous Universities and Institutes at a radius of less than 100 km, and the possibilities to host BSC and MSc students. But, it looks much easier than it was - and still is. We had to adapt to industry rules, strict deadlines, quick decisions and priorities, unexpected expenses, safety and employment law, plus the understanding and language of other disciplines, pharmacy, formulation specialists, technical, physical and theoretical chemists, and last but not least with patent lawyers." Continuous Focus on Innovation “Besides all, we continuously invest in research, and we host BSC and MSc students to establish a dynamic exchange with Universities,” Carsten says with pride. “But innovation does not only deal with scientific issues. Real success only comes from collaboration across different disciplines. This is possible only if all the common languages are spoken- of organic, bio, theoretical, and technical chemists, plus pharmacists, and, last but not least, of patent lawyers. Today, solid-state forms of crystalline polymorphs, co-crystals, salts, or amorphous states are our daily business. We can identify, select, or even produce different solid-state forms of APIs even when they influence or mutually counteract each other”. [1] The German Donor’s association (Stifterverband) is a powerful organization of 3’000 multinational companies, smaller enterprises and private persons with an annual funding volume of 150 mio EUR dedicated to science and education. Inolytix represents now SOLID-CHEM in Switzerland Are you fully aware that many essential physicochemical properties of an Active Pharmaceutical Ingredient (API) depend on its solid-state structure? Solubility, bioavailability at desired levels, shelf-life stability, hygroscopicity, compressibility, tableting ability, electrostatic charging, plus many others, can be crucial properties. And the decision is not trivial, which kind of solid-state form should be produced or selected for an API and which data are required by patent lawyers. Sophisticated analytical tools and the right expertise are essential for successful evaluations. Inolytix AG represents now SOLID-CHEM GmbH in Switzerland By working together on analytical projects, it became clear that SOLID-CHEM GmbH, Bochum, Germany possesses special equipment of instruments and the right expertise – especially for the pharmaceutical industry but also for others. Customers reacted very positively, for example, “…now my favorite CRO for ssNMR….”. Therefore, we are pleased to announce that Inolytix AG is now an exclusive partner in Switzerland for services offered by SOLID-CHEM GmbH. Offering a comprehensive service of solid-state analysis Exploring suitable solid-state forms (amorphous, polymorphs, salts, co-crystals, or solvates) requires a diverse methodological portfolio plus the expertise and the compounds for (co ) crystallization. Moreover, you need to precisely define all the appropriate analytical tools when matching your API with the final formulation. Many methods exist for determining solid-state forms because the intermolecular interactions responsible for the solid-state have a very subtle character. Diffraction- and thermochemical methods, particle determination, ssNMR, humidity determination, etc. - all this requires collaborating specialists overviewing such cross-networking analyses. Finally, they have to understand the different languages when speaking to organic, bio, theoretical, and technical chemists, to pharmacists, formulation specialists, and, last but not least, with patent lawyers. It will be our pleasure and ambitious task to provide you with the best analytical services for solid solutions for the solid-state of pharmaceutical products ranging from • pure APIs to complement tablet formulations • screening procedures of solid forms, • data for patents or stability tests Any questions about your solid product form? Contact us, and let’s discuss it! Please find below two supporting documents:
Inolytix, Aargau (Switzerland) - Inolytix is a startup launched in 2014 with the focus on accelerating product development and true understanding for the chemical and pharmaceutical industry. Through new and innovative analytical methods delivered by reliable and experienced partners such as SOLID-CHEM, Inolytix offers a customer-friendly service for R&D departments: providing the customer with a clear expert view and access to effective, but less known analytical methods, Inolytix manages the whole process from quotation to the industry- oriented report on the results.
Contact: Dr. Ralf Dümpelmann (for Switzerland), Founder and Managing Director [email protected], phone: +41 79 312 9327, www.inolytix.com SOLID-CHEM GmbH (Germany, Bochum) - Founded by Prof. Boese and Dr. Schauerte as a spin-off from the University of Essen, SOLID-CHEM GmbH has become an expert center of solid solutions for the solid-state of pharmaceutical products. The philosophy is, to support customers with expert opinions based on experimental, scientific, carefully retrieved data and on precisely documented experiments. Results and reports from investigations can be potentially used for litigations, and therefore, scientific integrity is our absolute priority. Contact: Dr. Carsten Schauerte, Managing Director and Co-founder, [email protected], phone: +49 234 936 90610, www.solid-chem.de  Surface analysis of solids as particles, fibers and plats can be very tricky. Dynamic gas adsorption with 15+ different gas probes, also known as Inverse Gas Chromatography (IGC), delivers very detailed information about surface energies, nanoroughness, polarity, acid/base properties (Lewis) and surface heterogeneity. Adscientis has 20 years of experience of analyzing samples by IGC with now over 800 different projects for the industry. Recently, Adscientis made a great step forward in IGC with a new automated system (neuronicIC) of high flexibility, accuracy and transparency of all process conditions overcoming thereby many shortcomings of existing instruments. Inolytix, founded in 2014, is an enthusiastic promoter of innovative analytical techniques and IGC in particular from the beginning. Analytical challenges of the chemical and pharmaceutical industry are successfully solved especially of solid and surface properties. With respect to IGC, the focus is on the german-speaking market, i.e. Germany, Switzerland and Austria. The annual organization of the International IGC-Symposium is one of the examples of community support and expert network. Inolytix and Adscientis start a global cooperation Now, with the new IGC instrument, both experienced companies decided to join forces for a global collaboration. This involves common communications about the use of IGC, the new instrument properties, application notes, webinars and an extension of the IGC Symposium worldwide. “It is exciting to work so closely together and to use our mutual understanding of surface characterization on a global scale”, says Ralf Duempelmann, Managing Director and Founder of Inolytix. Eric Brendle, CEO of Adscientis adds “After six years of some cooperation with Inolytix, it is now just the right time for a closer cooperation. We share the same spirit and enthusiasm to move IGC towards the next level”. Interested in surface and solids characterization? Having issues with qualities, batch-to-batch properties or understanding product modifications? Don’t hesitate to contact either one of the companies! Our expertise and their our willingness to characterize difficult surfaces will bring you forward – either using IGC or complementary analytical techniques. Adscientis, Wittelsheim (France) - Adscientis, founded in year 2000, is today recognized as the most reliable IGC specialist. With over 800 customer projects, the company acquired a unique set of skills and know-how on IGC techniques for the surface characterization of powders ranging from carbon black to pharma products, fibers and flat surfaces. The art of using dynamic adsorption and 15+ gas probes is refined continuously to deliver reproducible and quantitative values of surface energy, nanoroughness, polarities, acid/base properties, surface heterogeneity, specific surface areas and even HSP values. The results address crucial challenges as quality issues, product development and application behaviors. The development of an automated and highly flexible IGC instrument is based on customer needs and a result of the long-term practical experience.
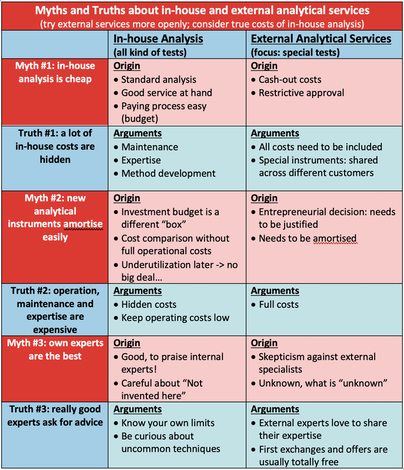
Contact: Dr. Eric Brendle, CEO, e.brendle(at)adscientis.com, phone: +33 389 4800042, www.adscientis.com Inolytix, Aargau (Switzerland) - Inolytix is a startup launched in 2014 with the focus on accelerating product development and true understanding for the chemical industry. Through new and innovative analytical methods delivered by reliable and experienced partners such as Adscientis, Inolytix offers a customer-friendly service for R&D departments: providing the customer with a clear expert view and access to effective, but less known analytical methods, Inolytix manages the whole process from quotation to the industry- oriented report on the results. Contact: Dr. Ralf Dümpelmann, Founder and Managing Director, [email protected], phone: +41 79 312 9327, www.inolytix.com 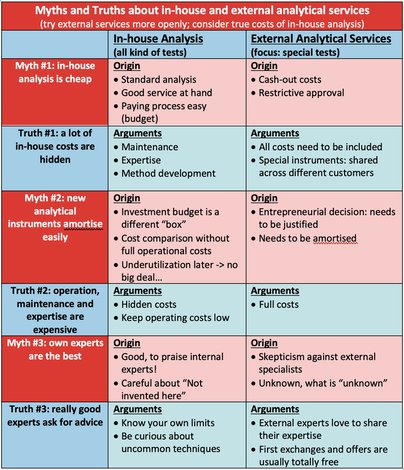 I have worked for 20 years in the R&D of global chemical companies and I always appreciated their own, in-house analytical services. They are fast, have known methods, all costs are budgeted and best: costs appear only somewhere in the depths of SAP. Great! Today, I’m running Inolytix Ltd and we bring innovative, less known analytical tools as external services into chemical companies. Why? It’s my believe and experience that many opportunities in understanding your F&E or quality issues are missed, simply because the known analytical methods are too often not the best. Or to quote Donald Rumsfeld “…there are also unknown unknowns, things we do not know we don't know.” In this article, I’d like to discuss the advantages of in-house analytical services, the advantages of external analytical services and how to balance both in the best possible way. To be a bit provocative, I’m stating some common believes as myths and explain the truth behind. Are your experiences different or do you share similar stories? Would be great to know ! Myth #1: in-house analysis is the cheapest option Yes and no. For standard analysis like HPLC, being used on a daily basis, hardly anything can beat your own analytical laboratory. It is not only comparable in costs to the cheapest external laboratory (even though some outsourcing labs argue on price), but also fast and results can be easily discussed with the experts in-house. But for analytical methods not being used every day (like e.g. Raman-FTIR, XPS or IGC), things look very different: These analytical services (if available in-house) seem cheap too but in fact, they are not. Truth #1: a lot of in-house costs are hidden Three factors seem to hide some of the true costs: First, service, maintenance and repair of analytical equipment is a considerable cost factor especially if not used on a regular basis. And these are cash-out costs. Second, method development for rarely used instruments are often difficult and time consuming. However, the analytical department needs to promote special methods, otherwise they will never be used – so costs are distributed. Least obvious, but very important is the third point: the value of expertise. Experts need exercise, many different samples and challenges. This is easy with daily analysis, but difficult with less used ones. Either the expertise is not very high or regular practice is costly – either way, this is not satisfying. Myth #2: new analytical instruments amortize easily NO – at least in many cases. We all know this justification: the new instrument costs e.g. EUR 150’000. With 150 samples over 2 years, costing EUR 1’000/sample externally pay-back time would be 2 years only: a convincing argument! But really?? Before coming to Truth #2, I would like to mention a fact which makes it indeed easy to invest into new equipment – regardless of any follow-up costs. The investment budget for a new instrument is simply a different “box” than the operational budget. It is common sense to allocate some money for new instruments. However, this investment budget is often being fully used, irrespective of whether or not the new instrument is indeed useful and cost efficient. Truth #2: operation, maintenance and expertise are expensive A true pay-back time calculation needs to incorporate all operating costs such as working time plus maintenance, method developments, amortization and the development of the required expertise to actually use the given instrument. The more underutilized the instrument is the worse becomes the calculation due to an increasing part of fixed costs. Let’s assume the in-house operating costs of the case mentioned above can be brought down to EUR 500 /sample. In this case, pay-back time doubles to 4 years! Some details are provided in an example including an FTIR instrument of USD 20’000 investment and external costs of USD 10’000. In conclusion, realistic pay-back times of analytical instruments are often much higher than anticipated. External services appear costly at first sight but in fact, they are often highly cost-efficient if compared on a 1:1 basis. Myth #3: in-house experts are the best Surely, in-house experts are often very good, especially with methods they always use. But what about the other methods? Last year, I have visited a company sophisticated on rheology using rotary viscosimeters. But they did not know about micro-rheology offering new possibilities in their case. It needs a good balance between the NIH-syndrome (not-invented-here) and curiosity plus openness for new methods. Truth #3: really good experts know when to ask for advice The solution for your analytical challenge may be in-house, but it might be also outside. Your experts are really good if they are curious, reach out to other experts and help to find the best analytical solution for you– and not just the one available. The first steps of external expertise are often free anyway. A short email or a phone call and you have more options to evaluate already. Surprisingly, I’m receiving these requests more often from development chemists than from analytical people. In conclusion, there are many rational arguments to be more open towards external analytical services – to save money and to increase the understanding of the product properties. Recordings of the presentations can be watched now.When we had decide because of Covid19, if the annual IGC Symposium should take place in 2020, we did not expect this successful outcome. Instead of doing it onsite in person, we took the opportunity to offer it online. And suddenly the number or registrations increased rapidly. At the end we had more than 150 registrations from all over the world. Another benefit of going online are the recordings that you can now watch afterwards, even if you did not attend the symposium. We published all those that we are allowed to publish on youtube. If you are interested in learning more about the applications and special details of inverse gas chromatography, you find the recordings below. If you are interested in the slides you can download them here: IGC Symposium 2020 - Recordings20 years of IGC experiences and learnings - |
|||||||||||||||||
|
We are eager to support you.
|





 RSS Feed
RSS Feed